Stem Cell Therapy for High Blood Pressure / Hypertension
Stem cell therapy offers new hope in the treatment of hypertension, a condition that affects millions of people worldwide and is a major risk factor for cardiovascular diseases....
5/8/20242 min read
Stem Cell Therapy Offers New Hope in Hypertension Treatment
Stem cell therapy offers new hope in the treatment of hypertension, a condition that affects millions of people worldwide and is a major risk factor for cardiovascular diseases. Preclinical research shows that mesenchymal stem cells (MSC) can lower blood pressure through mechanisms such as reducing inflammation, repairing kidney function, and modulating the sympathetic nervous system. This article discusses the potential of stem cell therapy for hypertension, supported by data from recent journal studies, although most are still at the animal stage and require further clinical trials in humans.
Mechanism of Action of Stem Cells in Hypertension
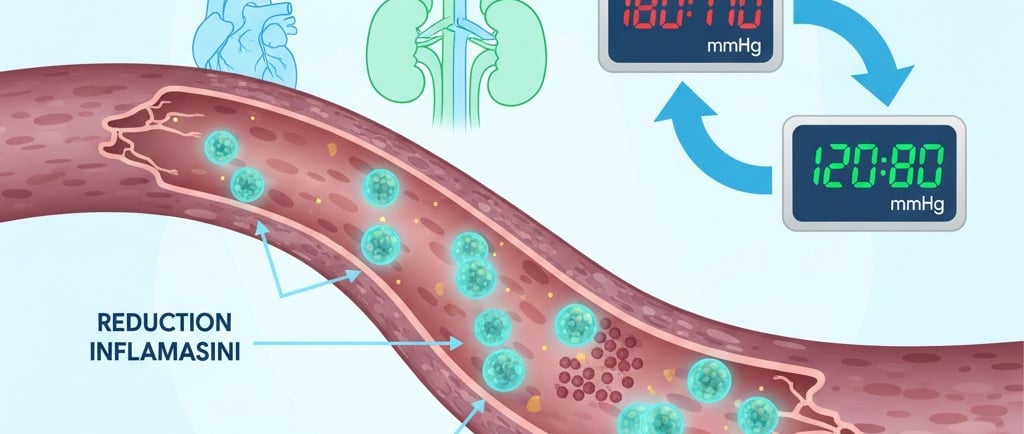
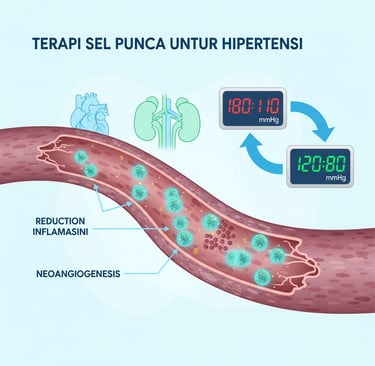
Mesenchymal stem cells, derived from bone marrow or induced pluripotent stem cells (iPSC), work by dampening inflammation and repairing blood vessels damaged by chronic hypertension. In a 2-kidney-1-clip (2K-1C) renovascular hypertensive rat model, the infusion of MSC at a dose of 2x10^5 cells via the tail vein in weeks 3 and 5 prevented the progressive increase in systolic blood pressure (SBP), resulting in significantly lower values compared to the control group (p<0.05). Another study on spontaneously hypertensive rats (SHR) showed that hiPSC-MSC accumulated in the spleen, releasing glutamate that activates sympathetic nerves in the spleen, increasing ChAT+ (choline acetyltransferase-positive) cells that produce acetylcholine as a vasodilator, thereby lowering blood pressure and inflammation in target organs. Additionally, MSC induces neoangiogenesis, reduces kidney fibrosis, and normalizes gene expression such as IL-1β, TNF-α, and angiotensin II (AT1) receptors, which are elevated in the medulla of stenotic kidneys.
Evidence from Animal Studies and Early Clinical Trials
Research on renovascular hypertension models shows that MSC enhance contralateral kidney function by maintaining natriuresis through a tubular-independent mechanism, where aquaporin-1 (AQP1) is suppressed in hypertensive kidneys, leading to increased urine flow and sodium excretion without altering the expression of major sodium transporters such as NHE-3, ENaC, or Na/K-ATPase. In a meta-analysis of 8 studies, MSC treatment significantly reduced SBP (SMD = -1.019, 95% CI -1.434 to -0.604, p=0.000), although mean arterial pressure did not change significantly. In early clinical trials for pulmonary hypertension (a specific form of hypertension in pulmonary arteries), autologous adult stem cells from patient blood reduced pulmonary arterial pressure from 41 mmHg (severe) to 24 mmHg (normal) after 9 months of follow-up in the first patient, with a similar pattern in other participants. Other studies reported reductions in SBP, decreased proteinuria, and detection of MSC in the renal cortex/medulla as well as the CNS, indicating the ability of cells to cross the blood-brain barrier to reduce sympathetic hyperactivity.
Challenges and Future Prospects
Despite its promise, stem cell therapy for essential systemic hypertension remains limited to preclinical studies, with increased mobilization of natural MSC in hypertensive patients related to left ventricular hypertrophy (LVH). Challenges include optimizing the administration route (intravenous, intratracheal, or direct implantation), genetic modification for stronger effects like prostacyclin synthase, and validation of advanced phase clinical trials for long-term safety. Regenerative clinics report stable blood pressure reduction 3-5 months post-fetal stem cell transplantation, with 85% of patients reducing or discontinuing antihypertensive therapy. Future prospects include combinations with conventional drugs for synergistic effects, especially in resistant hypertension, with a need for further research on mechanisms such as the influence of gut microbiota or epigenetics. Overall, this data supports the development of stem cell therapy as an innovative strategy to manage difficult-to-control hypertension.
